Decoding Viruses to Deliver Vaccines: Inside 360biolabs’ Bioanalysis Edges

360biolabs' 10th anniversary
The 10 Series: A decade of enabling future medicines
Over the past decade, 360biolabs has become Australia’s most comprehensive bioanalytical laboratory.
Powered by deep expertise in virology and immunology, our scientific teams have over 225 infectious disease studies and analysed more than 115,000 clinical samples, accelerating vaccines, antivirals and viral-vector therapies from development to patients.
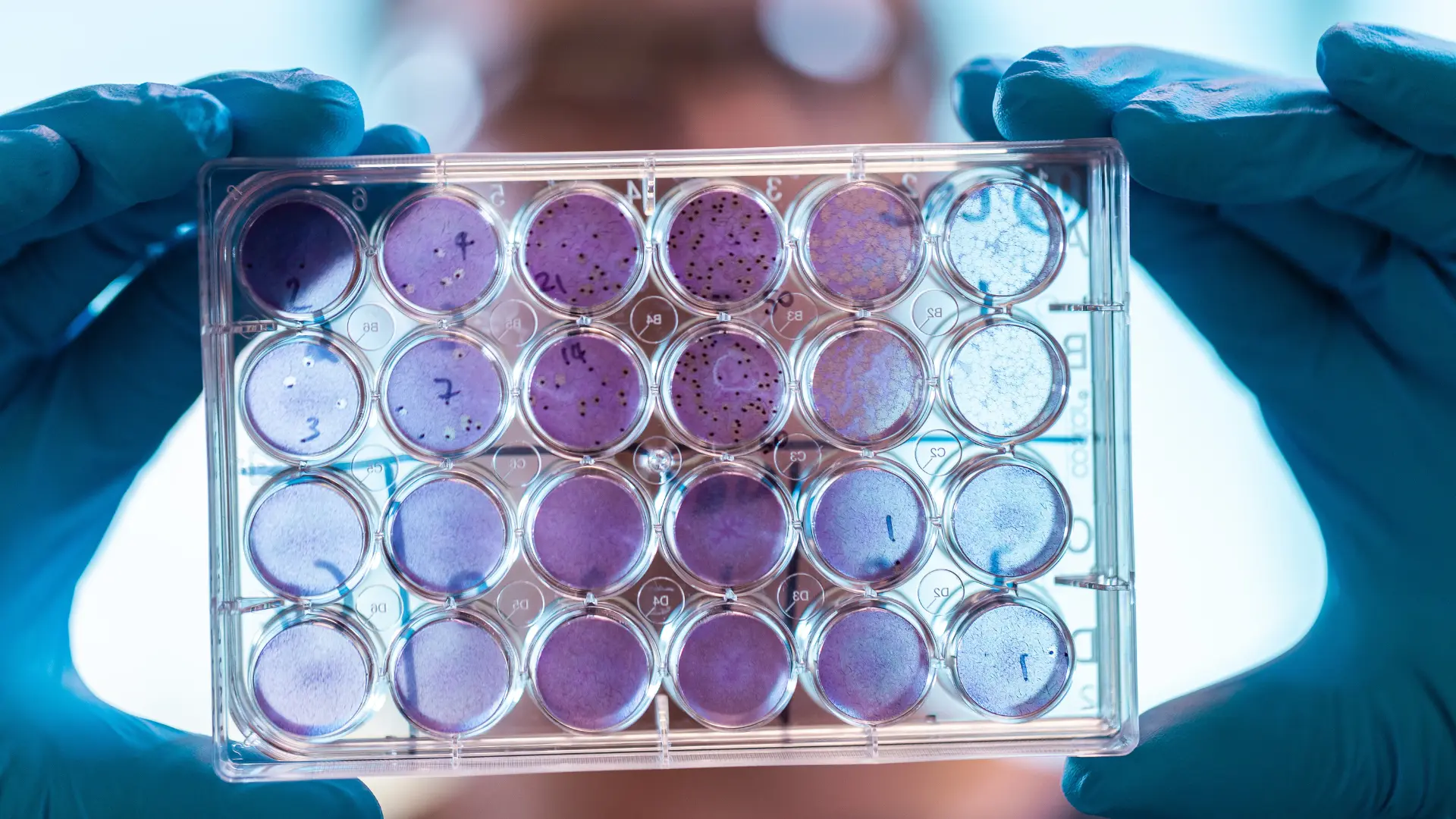
Foundations in Virology and Immunology with a Focus on Advanced Modalities
360biolabs is defined by deep scientific expertise in virology, immunology, and molecular biology. From the outset, we have focused on complex bioanalytical assays, including neutralisation tests (MNT), ELISAs, cell-based infectious disease assays, and advanced molecular platforms.
Our LC-MS/MS capabilities enable sensitive and specific detection of lipid nanoparticle (LNP) components to support the development of nucleic acid-based therapeutics, including mRNA vaccines. Using targeted mass spectrometry methods, individual lipid constituents, such as ionisable lipids, phospholipids, cholesterol, and PEGylated lipids, can be accurately quantified in complex biological matrices.
These assays support pharmacokinetic, biodistribution, and stability studies by providing precise measurement of LNP-associated lipids in plasma, tissues, and other sample types. Methods are developed and validated to ensure high sensitivity, selectivity, and reproducibility, with workflows optimised for low-level detection and robust performance.
This capability enables detailed characterisation of LNP formulations and in vivo behaviour, supporting regulatory submissions and advancing next-generation RNA and gene-based therapeutics.
This integrated capability, bringing multiple scientific disciplines together within a single laboratory has enabled the delivery of highly specialised, end-to-end bioanalytical solutions for infectious disease and vaccine development for a diverse range of modalities.
As demand has grown across international markets, laboratory operations have expanded to include certified BSL-2 and BSL-3 facilities in Melbourne and BSL-2 facilities in Brisbane, supporting safe and rigorous research across a wide spectrum of viral pathogens.
Today, capabilities span both established and emerging pathogens, including RSV, SARS-CoV-2, influenza and pandemic influenza strains, PIV, hMPV, adenovirus, measles and rubella, poxviruses, VZV, Zika, and EV71. We have introduced both live-virus assays plus pseudovirus assays where live-virus is not possible due to containment level or importation regulations. Our work extends to infectious disease and immunotherapy programs targeting pertussis, malaria, and peanut allergy.
This breadth reflects not only the scientific versatility of 360biolabs, but also its ability to rapidly respond to emerging infectious disease threats, meeting the growing global demand for high-quality bioanalytical support across infectious disease and immunotherapy.

High-Quality Assays for Next-Generation Vaccine Development
Our core strength lies in the design, optimisation, and validation of assays that characterise immune responses across diverse vaccine modalities. As platforms such as mRNA and viral vectors continue to advance, assay strategies are tailored to each program’s mechanism of action, immunogenic profile, and clinical endpoints, delivering robust, decision-ready data through clinical development.
At 360biolabs, we support next-generation vaccine development through a broad portfolio of scientifically rigorous assays designed to characterise immune responses, viral growth and delivery systems across different vaccine modalities:
Culture based assays:
To understand the efficacy and safety of antiviral drugs and vaccines through methods like Qualitative Culture (flask), and Quantitative Culture (TCID50 or Plaque Assay).
Viral neutralisation assays:
To determine the ability of antibodies to inhibit the infectivity of a virus, and characterise the immune response to vaccines through Plaque Reduction
Neutralisation Test (PRNT), Microneutralisation (MNT), and Focus Reduction
Neutralisation Test (FRNT).
Enzyme-linked immunosorbent spot (ELISpot):
To measure how many immune cells are activated by a vaccine by counting single cells that release cytokines, letting us measure specific T-cell and B-cell activity.
Hemagglutination inhibition (HAI) assays:
To measure antibodies that can block viruses like influenza from binding to cells to help show whether a vaccine triggers protective immunity, alongside other readouts such as cellular responses.
Cellular Responses:
With measurement through Immunophenotyping & Intracellular Cytokine Staining (ICS) techniques to show how T-cells and B-cells react to a vaccine and help determine whether the immune system is responding as expected.
Molecular assay:
By techniques to assess, gene expression via qPCR, dPCR, and NGS, mutation detection, gene panels, pathogen detection, and the measurement of viral vector titres and shedding allow us to understand what is happening at the genetic level during a vaccine response.
Biomarker assays:
To quantify both the strength and quality of vaccine-induced immune responses by measuring antibodies, cytokines, and other immune signals across sample types including serum, plasma, mucosal fluids, and saliva. These analyses are performed using advanced platforms such as MSD-ECL, bead-based luminescence systems, SIMOA (Single Molecule Array), and absorbance-based technologies, in both single-plex and multiplex formats.
Immunoassays:
By quantify vaccine-induced antibody responses using vaccine-specific ELISAs and serological testing for IgG, IgM, and IgA, enabling precise measurement of antibody levels and assessment of immune response.
Peripheral Blood Mononuclear Cells (PBMCs):
With isolated using systems such as Ficoll, SepMate and CPT, ensure high-quality cells for downstream assays including ELISpot, immunophenotyping and ICS. These assays are essential for analysing memory T-cells, B-cells, cytokine production and other immune responses elicited by a vaccine.
LC-MS/MS:
With capabilities enable sensitive and specific detection of lipid nanoparticle (LNP) components such as ionisable lipids, phospholipids, cholesterol, and PEGylated lipids.
Off-the-Shelf Assays and Vaccine Program Readiness
360biolabs offers a suite of validated, off-the-shelf assays designed to accelerate vaccine development. These ready-to-deploy platforms allow rapid immune monitoring, supporting early-stage screening through to clinical studies. By maintaining these assays in a state of vaccine program readiness, we can quickly adapt to new vaccine candidates and emerging pathogens, providing timely, high-quality bioanalytical data to drive decision-making.
This virus suite includes microneutralization assays for SARS-CoV-2, and RSV A and B strains, PRNT50 assays for metapneumovirus and parainfluenza and microneutralization and HAI assays for seasonal and pandemic influenza strains.
Where high-containment pathogens cannot be imported or cultured in Australia, such as influenza H5N1 or monkeypox, we have expanded our capabilities to include pseudovirus assay systems that mimic viral entry without full viral replication. These BSL-2 compliant models provide a safe and powerful way to measure neutralising antibodies and evaluate immune responses for pathogens that otherwise cannot be tested in BSL-2 or BSL-3 environments.
Seamless Global Sample Handling
COVID-19 highlighted just how efficiently global sample shipping can work. Between 2020 and 2024, 360biolabs supported over 25 SARS-CoV-2 clinical studies, including Phase 1 and 2 trials in Australia and multiple Phase 3 programs internationally. During this period, we received more than 60,000 samples from around the world, demonstrating that partnering with a highly experienced, quality-focused Australian bioanalytical laboratory can be smooth, reliable, and fully equipped to handle complex, large-scale vaccine programs.
360biolabs, Your Partner for the Australian Advantage
Australia has become an increasingly attractive destination for vaccine development. 360biolabs observed 80% growth in non-COVID vaccine clinical trials during the years 2020 to 2025, supporting over 90 vaccine clinical studies during this period. The trends for Q1 2026 indicate that Australia is continuing to attract large numbers of vaccine clinical studies with 2026 growth on track to exceed 100% compared with 2025.
Over this past decade, international biotech companies have increasingly launched vaccine programs in Australia, drawn by faster regulatory approvals, globally trusted data, and cost efficiencies that accelerate development timelines and attract investor support.
Respiratory viruses follow predictable seasonal patterns, winter peaks for influenza and RSV, late winter to spring for hMPV, type-specific peaks for PIV, and autumn-spring surges for HRV. This makes Australia an ideal location for vaccine trials, allowing studies to run during active virus circulation rather than waiting for the next season. By aligning programs with these transmission windows, developers can accelerate timelines, generate critical data faster, and reduce financial risk.
Australia’s R&D tax incentive can cut trial costs significantly, making Australia up to 60% more cost-effective than comparable studies in the US or Europe. As an OECD country operating under ICH-GCP standards, Australia also ensures that locally generated data is fully accepted by global regulatory authorities.
Looking Forward
Infectious disease research is evolving at a pace, requiring bioanalytical partners that can not only keep up with innovation but help drive it.
As capabilities continue to expand, through advanced assay platforms, enhanced laboratory infrastructure, and deepening scientific expertise, 360biolabs is well-positioned to support the next generation of vaccine and infectious disease clinical trials.
With a continued focus on emerging pathogens and complex therapeutic modalities, our mission remains clear: to deliver high-quality, decision-ready data that enables future medicines worldwide.
Interested in conducting a clinical trial in Australia with 360biolabs bioanalytical excellence?
